











USP42第二增補版新增了章節,增加了一個特定微生物的測試。現將中英文推送如下。
INTRODUCTION 介紹
The tests described in this chapter will allow determination of the absence of Burkholderia cepacia complex(Bcc),which can be detected under the conditions described.
本章節所描述的測試是允許確定BCC 的存在,可在描述的情況下檢測。
The tests are designed to determine whether a substance or preparation complies with an established specification for microbiology quality and/or to evaluate whether products ----especially those for inhalation use or aqueous preparations for oral,oromucosal,cutaneous,or nasal use----contain members of the Bcc.
該測試旨在確定一種物質或製劑是否符合已建立的微生物質量標準,並/或評估產品——特別是用於吸入或口服、口服粘膜、皮膚或鼻腔使用的含水製劑——是否含有BCC。
GROEITH-PROMOTING AND INHIBITORY PROPERTIES OF THE MEDIA AND
SUITABILITY OF TESTS FOR ABSENCE OF Bcc
培養基促生長和抑製作用以及BCC 方法適用性
Test each batch of ready-prepared medium and each batch of medium prepared from either dehydrated medium or ingredients.
測試每一批預製的培養基,以及每一批由脫水培養基或原料製成的培養基。
Preparation of Test Strains 測試菌株的準備
Use standardized stable suspensions of test strains(see Table1)NMT 5 passages removed from the original strain culture.
使用標準的穩定的測試用菌懸液,從原始菌株不超過5 代。
Table1.Test Strains of Microorganisms for Growth Promotion and Suitability Testing
表1 促生長及適用性測試的菌株

Microorganism 微生物
Grow each of the test strains separately in Soy-Casein Digest Broth or on Soybean-Casein Digest Agar at 30℃-35℃ for 18-24h.
分別在TSB/TSA 中培養上述每一個菌株於30℃-35℃ ,18-24h.
Use Buffered sodium Chloride-peptone Solution pH7.0 or Phosphate Buffer Solution pH7.2 to make the test suspensions.Use the suspensions within 24h if stored at 2℃-8℃.If purchased,follow the supplier’s introductions. If self-prepared cultures are used,follow a validated procedure (such as in Microbial Enumeration Test)for preparation. Use a challenge inoculum of NMT 100 colony-forming units(cfu) for growth promotion and suitability testing.
使用pH7.0 氯化鈉蛋白腖緩衝液或pH7.2 磷酸鹽緩衝液製備菌懸液。若保存在2o-8o 在24 小時內使用菌懸液。若是采購,按照供應商的說明書。如果使用自製培養物,請遵循經過驗證的程序(如微生物計數試驗)進行製備。使用不超過100cfu的挑戰菌進行促生長試驗和適用性試驗。
NEGATIVE CONTROLS 陰性對照
Include a negative control to verify the testing conditions. There must be no growth of microorganisms. A negative control is also performed when testing the products as
described in Testing of Products.
為確認測試條件需包含陰性對照。必須無微生物生長。在產品測試中描述的在產品測試過程中也需要進行陰性對照。
TEST FOR GROWTH-PROMOTING PROPERTIES, SOLID MEDlA
固體培養基的促生長試驗
Perform the Surface-Spread Method (see Microbial Enumeration Test, Growth Promotion Test, Suitability of the Counting Method and Negative Controls, Suitability of the Counting Method in the Presence of Product, Recovery of Microorganisms in the Presence of Product, Plate-Count Methods), inoculating each plate with a small number(NMT 100 cfu) of the appropriate microorganism (see Table 2). Incubate at the special temperature for NMT the shortest period of time specified in the test. Growth of the microorganism comparable to that previously obtained with a previously tested and approved batch of medium occurs.
采用塗布法(見微生物計數法,促生長試驗,計數方法適用性和陰性對照,產品存在時的計數方法適用性,產品存在時的微生物回收率,平皿法),接種不超過100cfu 的合適的微生物(見表2)。在特定的溫度下培養不超過特定培養時間的最短時間。微生物的生長可與以前通過先前測試和批準的一批培養基獲得的微生物相媲比較。
Table2.Microoganisms for the Growth-Promoting,Inhibitory,
and Indicative properties of the Media
表2.用於培養基促生長、抑製和指示的微生物
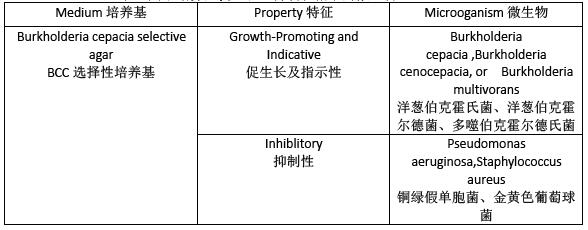
TEST FOR INHIBITORY PROPERTIES, SOLID MEDIA
固體培養基的抑製性測試
Inoculate the appropriate medium with at least 100 cfu of the appropriate microorganism. Incubate at the specified temperature for NLT the longest period of time specified in the test .Inhibition of growth of the indicated microorganisms occurs(see Table 2).
使用正確的微生物接種至少100cfu 至合適的培養基中。在特定的溫度下培養不低於特定培養時間的最長時間。抑製規定的微生物的生長(見表2)。
TEST FOR INDICATIVE PROPERTIES指示性測試
Perform the Surface-Spread Method (see Microbial Enumeration TestsGrowth Promotion Test, Suitability of the Counting Method and Negative Controls, Suitability of the Counting Method in the Presence of Product, Recovery of Microorganisms .In the Presence of Product, Plate-Count Methods), inoculating each plate with a small number(NMT 100 cfu) of the indicated microorganism. Incubate at the specified temperature for a period of time within the range specified in the test. Colonies are comparable in appearance and indication reactions to those previously obtained with a previously tested and approved batch of medium (see Table 2).
采用塗布法(見微生物計數法,促生長試驗,計數方法適用性和陰性對照,產品存在時的計數方法適用性,產品存在時的微生物回收率,平皿法),接種不超過100cfu 的合適的微生物(見表2)。在規定的溫度下,在試驗規定的範圍內培養一段時間。菌落在外觀和指示反應方麵與以前通過先前測試和批準的一批培養基獲得的菌落相當。
Suitability of the Test Method 測試方法適用性
The ability of the test to detect Bcc in the presence of the product to be tested must be established. The incubation time for the method suitability should not exceed the shortest incubation period specified. Suitability must be confirmed if there is a change in testing performance or a change in the product that may affect the outcome of the test.
必須建立在待測產品存在的情況下檢測BCC 的能力。方法試用性的培養時間不應超過規定的最短培養時間。如果測試性能發生了變化,或者產品發生了可能影響測試結果的變化,則必須確認適用性。
For each new product to be tested, perform the sample preparation as described in Testing of Products. At the time of mixing, add each test strain in the prescribed growth medium. Inoculate the test strains individually. Use a number of microorganisms equivalent to NMT 100 cfu in the inoculated test preparation.
對每一個要測試的新產品,按照產品測試中描述的方法進行樣品準備。在混合的時候,在規定的生長培養基中加入每個測試菌株。分別接種試驗菌株。在接種試驗製劑中使用一些相當於不超過100 cfu 的微生物。
Perform the test as described in Testing of Products, using the shortest incubation period prescribed. Bcc microorganisms must be detected with the indication reactions described in Interpretation.
按照產品測試中描述的方法,使用規定的最短培養時間進行測試。BCC 必須能檢測出在解釋中描述的指示性反應。
Any antimicrobial activity of the product necessitates a modification of the test procedure (see Microbial Enumeration Test, Growth Promotion Test, Suitability of the Counting Method and Negative Controls, Suitability of the Counting Method in thePresence of Product,
Neutralization/Removal of Antimicrobial Activity).
該產品的任何抗菌活性都需要修改測試程序(見微生物計數法,促生長試驗,計數方法的適用性和陰性對照,產品存在時的計數方法適用性,抑菌活性的中和/去除)。
TESTING OF PRODUCTS 產品測試
Sample Preparation and Pre-Incubation 樣品製備和預培養
Prepare a sample using a 1-in-10 dilution of NLT 1 g of the product to be examined.Use 10ml or the quantity corresponding to 1 g or 1ml to inoculate a suitable amount(determined as described in Suitability of the Test method) of Soybean-Casein Digest Broth or an appropriate dilution of Soybean-Casein Digest Broth as determined during method suitability (for example ,a 1:10 dilution may be required when conducting optional testing of pharmaceutical waters). Then mix and incubate at 30o-35ofor 48-72h.
用1:10 倍稀釋的不少於1g 的待測樣品製備供試液。用10ml 或相當於1g 或1ml的合適的(根據測試方法的適用性確定)供試液接種至TSB 或在方法適用性確定的適當體積的TSB 中(例如,在進行製藥用水可選檢測時,可能需要1:10 稀釋)。然後混合及在30℃-35℃ 培養48-72h.
Selection and subculture 選擇和繼續培養
Subculture by streaking on a plate of Burkholderia cepacia selective agar(BCSA),and
incubate 30o-35o for 48-72h.
在BCSA 上劃線繼續培養,30o-35o 培養48-72h。
Interpretation 解釋
The possible presence of Bcc is indicated by the growth of greenish-brown colonies with yellow halos, or white colonies surrounded by a pink-red zone on BCSA .Any growth on BCSA is confirmed by identification tests. See Microbial Characterization,Identification,and Strain Typingfor additional information.
BCC 可能存在的指示性特征為生長帶有黃色光環的綠棕色菌落,或在BCSA 顯示被粉紅色區域包圍的白色菌落。任何在BCSA 上的菌落需要通過鑒定測試確認。更多信息見微生物特性、鑒定和菌株分型.
The product complies with the test if clones of the types described are not present or if the confirmatory identification tests are negative.
如果所述類型的菌落不存在,或者確認性鑒定測試為陰性,則產品符合測試。
RECOMMEND CULTURE MEDIA 推薦的培養基
[Note-This section is given for information.][注:本節內容僅供參考。]The following solutions and culture media have been found satisfactory for the purposes for which they are prescribed in the tests in this Pharmacopeia . Other media may be used provided that their suitability can be demonstrated.
下列溶液和培養基已被發現符合本藥典測試規定的目的。如果能夠證明其他培養基的適用性,則可以使用其他培養基。
Stock Buffer Solution 儲備緩衝液
Transfer 34 g of potassium dihydrogen phosphate to a 1000-ml volumetric flask,dissolve in 500 ml of Purified Water adjust with sodium hydroxide to a pH of 7.2±0.2,add Purified Water to volume,and mix. Dispense in containers,and sterilize. Store at a temperature of 2o-8o.
將34 克磷酸二氫鉀轉移到1000 毫升的容量瓶中,溶解於500 毫升純淨水中,用氫氧化鈉調至pH 值7.2±0.2,加入純化水,攪拌均勻。分裝於容器內,並滅菌。儲存溫度為2℃-8℃。
Phosphate Butter Solution pH 7.2 pH7.2 磷酸鹽緩衝液
Prepare a mixture of purified Water and Stock Buffer Solution (800:1 v/v),and sterilize.
準備純化水和儲備緩衝液(800:1 v/v)混合,滅菌。
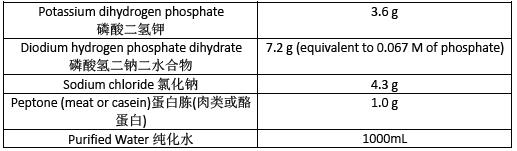
Buffered Sodium Chloride-Peptone Solution pH7.0 pH7.0 氯化鈉-蛋白腖緩衝液
Prepare Buffered Sodium Chloride-Peptone Solution pH 7.0 as directed in table 3.Sterilize in an autoclave using a validated cycle.
按表3 所示製備pH 7.0 氯化鈉-蛋白腖緩衝液。在滅菌鍋中使用經過驗證的程序滅菌。
Table 3 表3
Soybean-Casein Digest Broth 胰酪腖大豆肉湯
Prepare Soybean-Casein Digest Broth as directed in table 4 .Adjust the pH so that after sterilization it is 7.3±0.2 at 25o.Sterilize in an autoclave using a validated cycle.
按表4 製備TSB。調節pH 使得滅菌後在25o 為7.3±0.2.在滅菌鍋中使用經過驗證的程序滅菌。
Table 4 表4

Burkholderia cepacia Selective Agar 洋蔥伯克霍爾德菌選擇性瓊脂
Prepare BCSA as directed in Table 5. When preparing media in-house,first prepare the base ingredients without the antibiotics. Adjust the pH so that after sterilization it is 6.8±0.3 at 25o.Sterilize in an autoclave using a validated cycle.Cool the base medium to 45o-50o and add a 1% solution of the sterile filtered antibiotics ,mix,and pour into the plates.
按表5 製備BCSA。當在實驗室準備培養基時,首先準備沒有抗生素的基本成分。調節pH 使得滅菌後在25o 為6.8±0.2.在滅菌鍋中使用經過驗證的程序滅菌。將基礎培養基冷卻至45℃-50℃,加入1%的無菌過濾抗生素溶液,混合後倒入平板中。
Table 5 表5

上一篇:微生物計數試驗——營養和添加劑
下一篇:沒有了!

